Research in the Group is concerned with diverse aspects of the chemistry of carbohydrates and proteins. A multidisciplinary approach involving synthetic chemistry, molecular recognition methods (NMR, electrochemistry, calorimetry, etc.), and biophysics is followed to develop molecular sensors and drug carriers as well as to understand the mechanism of the binding interactions of proteins with bioactive molecules of therapeutic interest.
Cyclodextrins
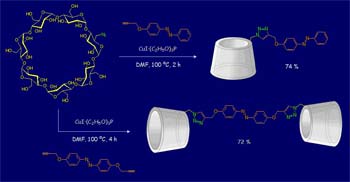
A part of our effort in synthetic organic chemistry is directed towards the development of cyclodextrin derivatives with functional and/or improved inclusion complexation properties.
We investigate synthetic methodologies that allow for the synthesis of functional β-CD, bis(β CD) and multi-β-CD systems.
Cyclodextrin-Based Sensors
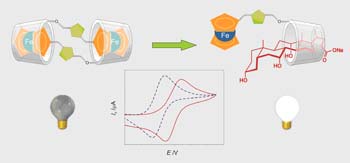
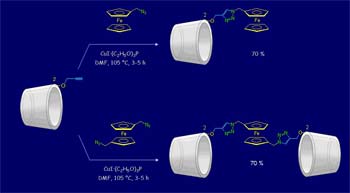
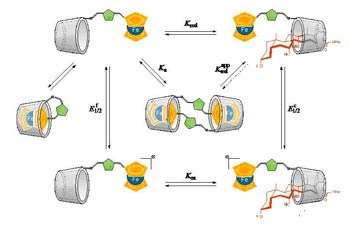
Ferrocene containing compounds with molecular recognition binding sites have received much attention in recent years due to the possibility of building redox-switching or -sensing systems of a molecular or supramolecular nature that can be controlled through the application of external stimuli.
Ferrocene features make it a good building block for the construction and design of redox molecular switches and sensors by conjugation with molecules involved in molecular recognition processes.
Cyclodextrin-Based Molecular Delivery Systems
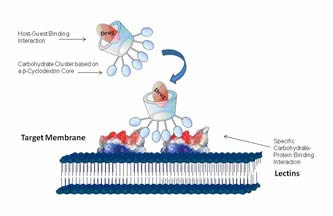
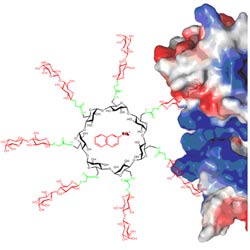
Clustered carbohydrates on simple scaffolds, surfaces, linear polymers or attached on the terminal groups of dendrimers, have been the focus of attention of many researchers, from synthesis to biological and pharmaceutical applications. This interest on glycoside clusters and dendrimers is based on the glycoside cluster effect or multivalent effect. Accordingly, clustered copies of carbohydrate ligands -multivalent ligands- interact with their protein receptors in a stronger and more selective fashion than the analogous single –monovalent- ligands. This phenomenon is ubiquitous in nature and involved in a wide range of important biological functions. If the glycocluster has the ability to encapsulate guest molecules, and particularly drugs, it could be possible to design a molecular delivery system with the potential of carrying drugs in a noncovalent way toward a specific biological receptor.
The use of CDs as a scaffold of cluster glycosides offers several advantages over other macrocyclic compounds: it is more readily available and affordable, more biocompatible, and has the ability to form inclusion compounds with a large variety of guests in aqueous solution.
As a part of this project we have synthesized several types of CD-based glycoclusters and studied their inclusion complexation properties and their binding abilities towards lectins.
Carbohydrate-Based Electrochemical Sensors
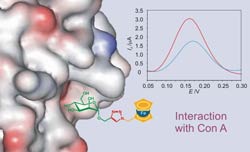
The covalent attachment of electroactive residues with molecules containing binding sites for molecular recognition is a way to construct redox switches and sensors, which can be controlled by external stimuli. The metallocene ferrocene meets the requirements to become a good building block for the construction of these molecular devices: ferrocene easily undergoes a monoelectronic and reversible oxidation which can be controlled both chemically and electrochemically, it is stable in water solutions and in most reaction conditions, and its synthetic approaches are well established. Given these facts, we investigate methods for the synthesis of monovalent and multivalent ferrocene-carbohydrate conjugates and study their abilities to form complexes with model receptors and lectins (Con A, PNA and galectins). The molecular recognition properties of the neoglycoconjugates and the electrochemical sensing abilities are studied by isothermal titration calorimetry, nuclear magnetic resonance and electrochemical methods.
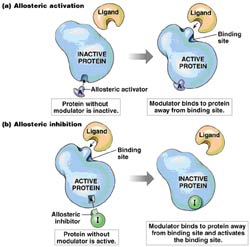
The role of each protein can be regulated through interaction with other proteins or with different types of low molecular weight molecules (small organic molecules or metals) known as ligands, whose function can be diverse and may act as activators, inhibitors or cofactors sometimes essential for biological activity.
In most cases, protein-ligand interactions are usually reversible.
The study of ligand-protein interactions also facilitates the understanding of processes of transport of exogenous substances, and establishes rules for the development of new drugs.
The main objective of our research is based on increasing knowledge of the thermodynamic factors involved in protein-ligand interactions. The development of this research combines biophysical and molecular biology techniques.
The characterization of intermolecular interaction is based on evaluating the specificity and strength of the interaction.
In addition, we analyze the influence of certain experimental variables such as pH, ionic strength, temperature, among others. In addition, the use of mutants in specific aminoacids increases the knowledge of the role of those residues in the catalysis and/or ligand binding.
Alternatively, spectroscopic and calorimetric techniques are used to carry out a global thermodynamic characterization.
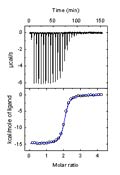
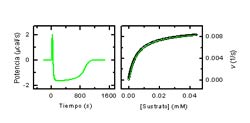
A comprehensive characterization of an intermolecular reaction involves the determination of the stoichiometry and the change in thermodynamic potential between the initial and final state: affinity, Gibbs energy, enthalpy and entropy of binding.
These techniques provide, therefore, a description of the interaction energy.
In addition, the change of experimental variables (temperature, pH, etc.) and the molecules involved it makes possible to obtain information about atomic interactions determinants in the binding process (hydrogen bonds, van der Waals, electrostatic, etc.).
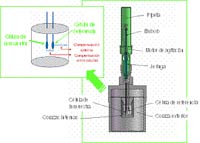
Calorimetric techniques are particularly suitable for the characterization of energy, because the direct observable is the heat of reaction variable that is present in any chemical reaction.
For this reason the calorimeters are called universal detectors.
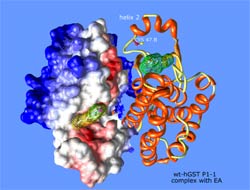
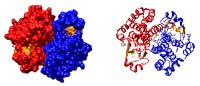
Our research group has performed the first calorimetric studies with the glutathione transferase enzyme. These studies have provided wide information on the binding mode of substrate (GSH) and some competitive inhibitors. The glutathione S-transferases are dimeric enzymes of approximately 25 kDa per subunit. In turn, each of these subunits consists of two distinct domains: an N-terminal and one C-terminal, the latter of α-helical character, counting each subunit, too, with at least two binding sites: one for the substrate GSH (G-site) and another site for the electrophilic substrate (H-site).
The human glutathione S-transferase (hGSTP1-1) is part of a family of dimeric multifunctional enzymes present in most forms of life, and belonging to Phase II of the metabolism of cytotoxic drugs.
The role played by these enzymes is encompassed within intracellular detoxification processes of harmful chemicals, catalyzing the nucleophilic attack of sulfur atom of the substrate GSH on electrophilic group on a second substrate (physiological or xenobiotic), forming glutathione conjugates, higher solubility in aqueous medium, which facilitates their excretion outside the cell.
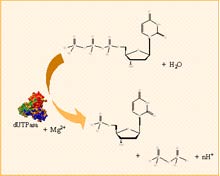
However, the detoxifying function of this enzyme plays a sometimes side effects during chemotherapy, since the GST has also been associated with effects of drug resistance by tumor cells. In fact, it has been found that certain forms of GST are expressed at high levels in tumor cells of mammals, so that the level of expression of the GST and its biochemical properties are crucial for determining cellular resistance to carcinogens, drugs antitumor, environmental toxins and products of cellular oxidative stress.The enzyme deoxyuridine 5'-triphosphate nucleotide hydrolase (dUTPase, EC 3.6.1.23) catalyzes the hydrolysis of α,β-pyrophosphate of dUTP to provide PPi and dUMP, using Mg2+ as cofactor.
This enzyme plays a key role in the integrity of DNA in cell division, preventing the accumulation of dUTP and ensuring at the same time, the provision of dUMP, the substrate of thymidylate synthase in the biosynthesis of dTTP.
However, scientific interest for dUTPasa enzyme goes beyond, and for example, the enzyme from Plasmodium falciparum, the protozoan parasite that causes human malaria, is being investigated for the development of cytostatic anticancer drugs.
The dUTPasa is an enzyme found in most living organisms. According to its quaternary structure, the known dUTPasas fall into monomeric, dimeric, or trimeric, the last two being the most studied.