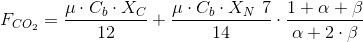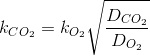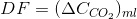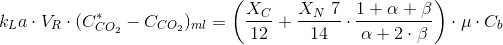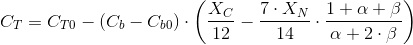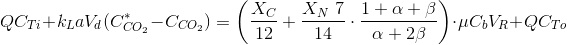4. CO2 and O2 exchange. Inorganic carbon equilibria.


4.4. CO2 transfer in photobioreactors..
This is the gist of the question. What CO2 rate do we need to supply to a microalgal culture? What mass transfer capacity (kLa, Vd) does that requires?
To answer this, we need to consider the particular circumstances of the cultures, as we do next.
-
•pH controlled by on-demand CO2 injection. Batch and continuous cultures.
As you probably remenber, to size the mass transfer capacity of a device, we need the required transfer rate (F, in mole per second), the driving force (C*-C, in mole per volume unit) and the volumetric coefficient kLa (1/t).
CO2 consumption rate.
Under constant pH, this is the uptake rate plus the CO2 needed to neutralize the alcainity released by the nitrate metabolism. The expression is shown next:
The condition of constant pH means that α and β are constant and known.
The first contribution represents the carbon incorporated to the biomass.
The secons contribution in the CO2 consumed to neutralize the alcalinity of the nitrate metabolism. The coefficient (1+α+β)/(α+2β) takes into account the part of the CO2 absorbed that dissociates to bicarbonate (neutralizing 1 OH-) or to carbonate (neutralizing 2 OH-).
The kLa for CO2 is more difficoult to measure than the for O2 but it can be estimater from this last one (transfer coefficient in the same system for different substances tend to be poportional to the square root.
Or, much better, from an adequate reference in literature.
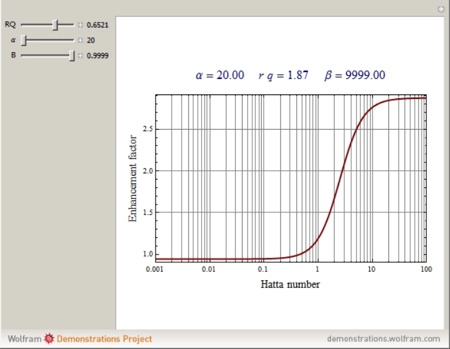
Additionaly, in alkaline conditions CO2 reacts with the OH- and diffusion stops being a bottleneck. In that case the absorption rate is accelerated by a ratio that is given by the "enhancement factor" which can be calculated from the Hatta number. This is studied in gas-liquid reactions. You can find more information about this here.
You can find more info about mass tranfer in gas-liquid systems with chemical reaction at Wolfram Alpha site. You can get an overview of the influence of the desigh and operating parameters on the Hatta number and thus on the chemical enhancement factor.
The absorption of CO2 in alkaline aqueous media is a second-order process.
For the driving force (DF), we will use the same definition as we did for oxygen: the diference between the equilibrium composition with the sparging stream (of known composition YCO2) obtained with the Henry Law (or whatever other means you see fit). Remember that you should need to do a mass balance to the aeration stream to check if its composition change. In this ocassion it will because we intend to transfer most of the CO2 to te culture. Thus, we will assume we will be using the logarithmic mean:
Batch cultures.
The transfer rate is to equal the uptake rate. This is constantly changing in a batch culture. The mass balance is:
VR is the volume of the degasser. It can be the size of the whole culture. To maximize the driving force, pure CO2 can be used. You should design for the maximum mass transfer needed. To calculate the free CO2 concentration in the culture we will need the total inorganic carbon, CT, as wee as the pH. The CT is:
Now you are all set to deal with this tipe of cultures. Remember that in this case pH is known as it is controlled by an automated device that injects CO2 when the culture so requires it.
Continuous cultures in steady-state.
Now the mass balance is somewhat more complicated as it needs an input and an output for the liquid phase. The variables are the same as in the last page: CTi and CTo are the inorganic carbon in the input and output phase, VR, reactor volume, Q, input and output flow rate. The mass balance for carbon is:
Since pH is constant and known, α and β are also known and the CO2 transfer rate needed is easily obtained from the equation above.
Note that this expression will give average transfer rates. In real systems, CO2, is in many cases injected discontinously.
Questions:
-
• Find the diffusivity of O2 and CO2. Find the square root of its ratio. This is the number that relates the mass transfer coefficient of these two gasses.
LITERATURE
-
• Other cases.
If pH is not kept constant, pH will change as CO2 is consumed and OH- released from nitrate metabolism. In this case you will need the full equations we presented in the last page. Be warned that the so-called "alkalinization rate" (the number of OH- released pr H+ taken up for every NO3-) may vary depending on several environmental conditions we are not still ready to deal with.
As for the engineering side, it is enough to take a conservative point of view and desigh for the maximum carbon uptake and CO2 absorption rate that we might expect to find.